Nex for Medtech
NEX: The Prebuilt Software Platform for MedTech
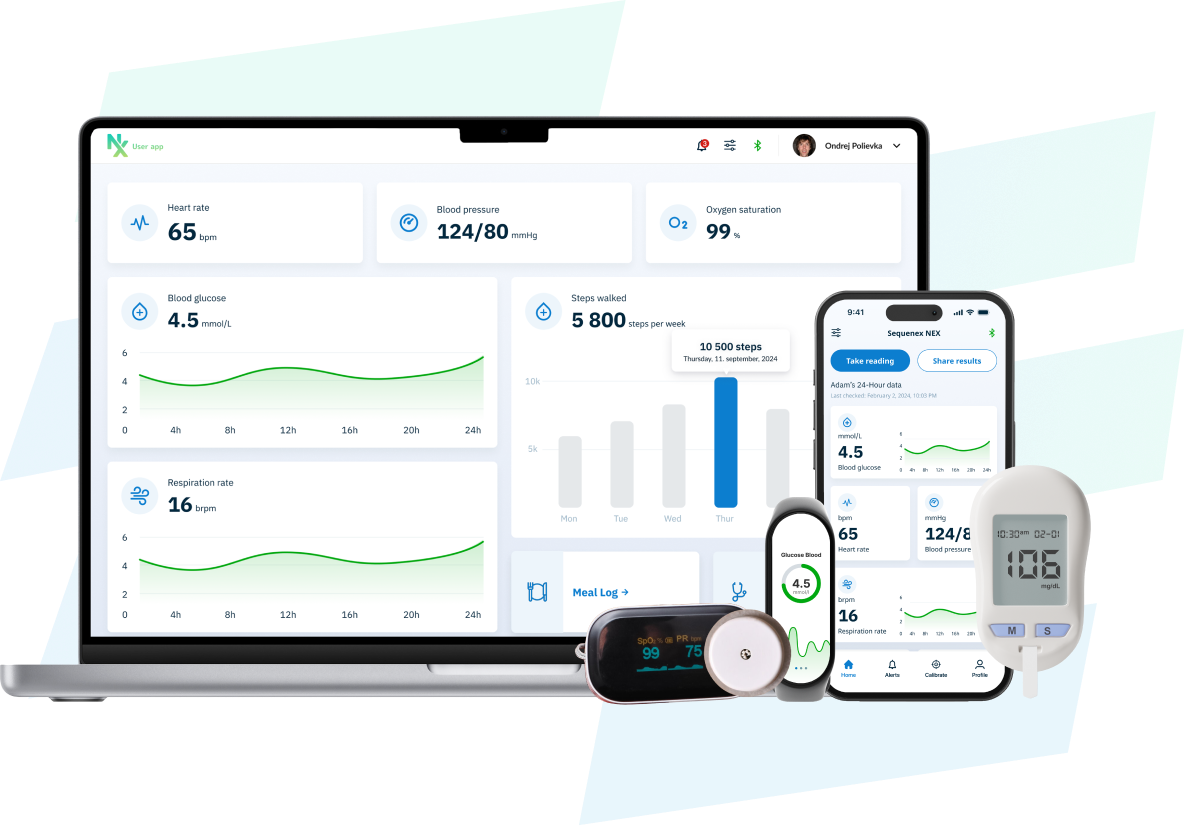
Focus on Your Device. Not the Software.
- Prebuilt and regulatory-ready for connected medical devices and SaMD. NEX provides a complete foundation: mobile apps, secure cloud services, dashboards, device connectivity, and administrative tools.
Real-Time Device Data...Fast
Capture CGM, biosensor, wearable, and connected device data through prebuilt mobile apps and cloud services. Support clinical studies, data collection, and early product validation.
Why Choose NEX?
Delivered Faster. Owned by You.
Built for Regulated Medical Devices
Developed under an ISO 13485-certified QMS in accordance with IEC 62304 and ISO 14971.
Each implementation includes documentation structured for inclusion in your Design History File (DHF).
Start Ahead, Not From Scratch
Don’t build your software from scratch.
NEX provides a prebuilt starting point so you can focus on what differentiates your device: Your Data, Algorithms, and User Experience.
Extend what’s already in place instead of building from zero.
Owned by You
NEX is deployed in your environment, giving you full control over your product roadmap, regulatory strategy, data, architecture, and long-term evolution.
Engineered for Acceleration.
NEX combines a prebuilt foundation with AI-assisted workflows to accelerate development, testing, and documentation.
All outputs are reviewed under structured design controls to ensure quality and regulatory rigor.
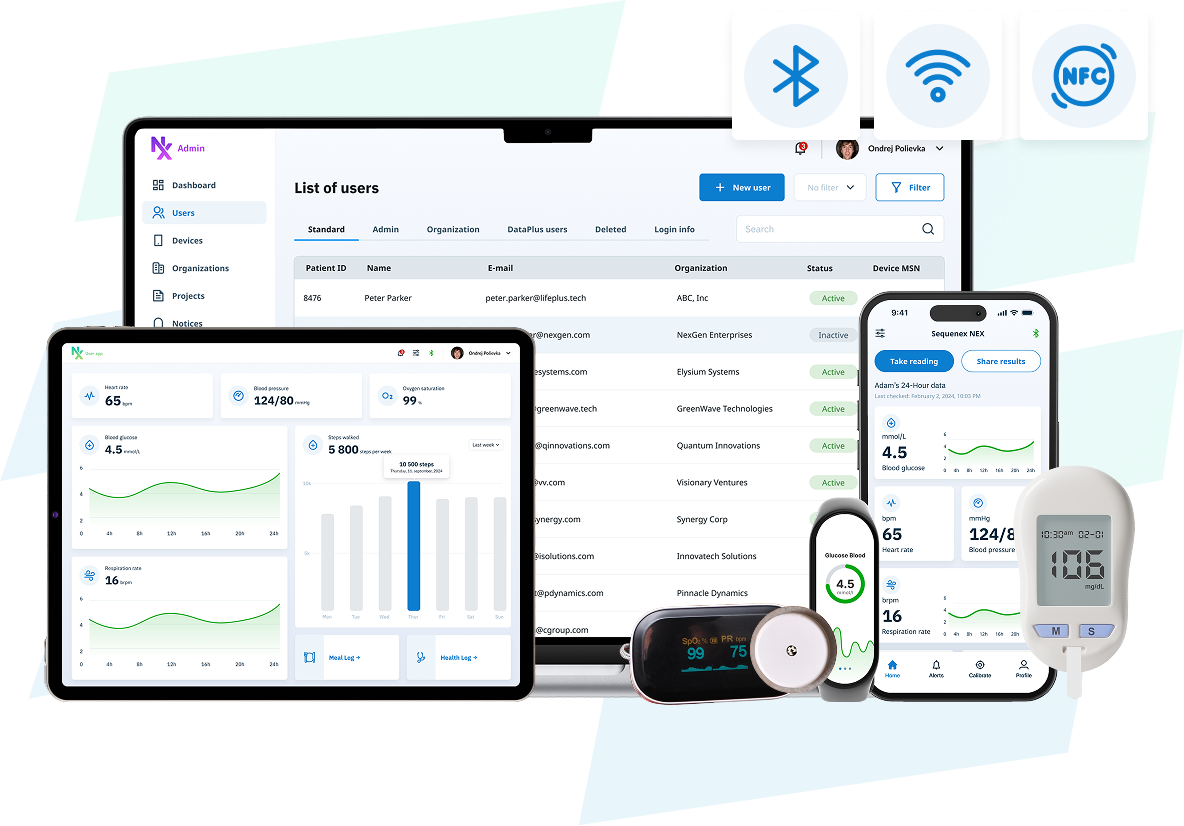
One Foundation: from MVP to Commercial
Prototype
Start collecting real device data immediately using a production-grade foundation. No "throwaway" prototypes.MVP
Extend this same foundation to support user workflows, validation, and investor-ready demos without rebuilding your data layer.Pre-Clinical & Clinical
Use the same foundation to support preclinical validation, feasibility, and IDE clinical studies with consistent data and traceability.Commercial
Confidently scale to production/commercial deployment. No migrations. No re-architecture. Confidently accelerate your time to market.
Rapid Time to Market
Prebuilt and Customizable iOS & Android Apps, Secure Data Cloud, Dashboards, and Admin Portal: Start with a fully functional foundation. Configured and extended to fit your device’s unique requirements.Full Service Software and Support
NEX provides a complete, prebuilt foundation for your product—extended and configured to your device and use case. Our team supports implementation, integration, and ongoing evolution so you can move forward without rebuilding your software.
Secure & Compliant Cloud Infrastructure
Secure infrastructure for healthcare applications, with encrypted data handling, access controls, audit logging, and resilient backup and recovery.
Designed to support HIPAA and GDPR requirements where applicable.
ISO 13485 Certified QMS
NEX is developed and maintained under an ISO 13485–certified Quality Management System, with controlled design, verification, validation, and change management processes.
Design History File (DHF) Ready
Each implementation includes structured documentation for your Design History File (DHF), including requirements, traceability, V&V, risk artifacts, and SBOM.
Design Controls
Software development follows IEC 62304 with risk management under ISO 14971—ensuring traceable, controlled, and repeatable development.
Built-In Compliance Features
System-level controls including role-based access, audit trails, and electronic record management—supporting compliant operation of your product.
Designed to support 21 CFR Part 11 requirements where applicable.
Customization and Flexibility
Configurable UI and Features
Easily customize the platform’s user interface and functionalities to suit your specific needs and branding.Adaptable Architecture
Modify and expand the platform to meet the unique requirements of your device.
A True Product Development Partner
NEX + Sequenex Engineering
NEX provides the foundation. Sequenex engineering configures, extends, and delivers product software for your device.
Controlled Change Management
Enhancements and modifications are executed under structured design controls with impact analysis, testing, and documentation.
Regulatory Continuity
Maintain design history, risk management, and lifecycle documentation as your product evolves. We work with your Quality/Regulatory.
Scalable Product Development
Expand your product using an established foundation, with engineering support as needed—without rebuilding or overextending internal teams.
Cost and Resource Efficiency
